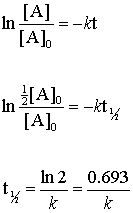half life formula for first order reaction
T 12 0693k. Integrated Rate Law Equation for Zero Order Reaction.
2λ 2 0693.

. T ½ 1 k A o Top. For a first-order reaction the half. The half-life equation for a first-order reaction is t12ln2k t 1 2 ln 2 k.
Half Life of First Order Reactions First-Order Reactions We can derive an equation for determining the half-life of a first-order reaction from the alternate form of the integrated rate law as follows. Justify from the rate equation for first-order reactions that the half-life period for such reaction is independent of the initial concentrations of the reactants. 2 k t 1 2.
As for other reaction orders an equation for zero-order half-life may be derived from the integrated rate law. Converting a half life to a rate constant. Here is how the Rate constant of first order reaction calculation can be explained with given input values - 0040547 ln0303-0110.
Equations for Half Lives. Get the free Half Life Calculator first order reaction widget for your website blog Wordpress Blogger or iGoogle. It is a constant and related to the rate constant for the reaction.
Numericals on zero. The half-life of a first-order reaction is given as t 12 0693k. It is denoted by t12.
Ln 12A0A0 -kt12. Half-time or half-life of a first-order reaction is the time taken to reduce the concentration of the reactant to half of its initial value. In each case we halve the remaining material in a time equal to the constant half-life.
A A 0 e k t AA_0 e-kt A A 0 e k t. How do you find the half-life of a second-order reaction. Find the time it would take for a.
Given that for a First Order reaction the half-life is twice the value of the rate constant find the value of the rate constant of the reaction. λ 0. Using the half-life equation derived from the concentration-time equation as shown in example 1 we can solve for the initial concentration of reactant.
Loge A0 A At half life period t t12 and A A02. T 12 0693k. FracR_0R From the definition of the half-life of a first-order reaction at t t 12 and R R 0 2.
Then write the half-life equation as. T ½ A o 2k For a first order reaction A products rate kA. Hence the half-life of a first order reaction is given as the following.
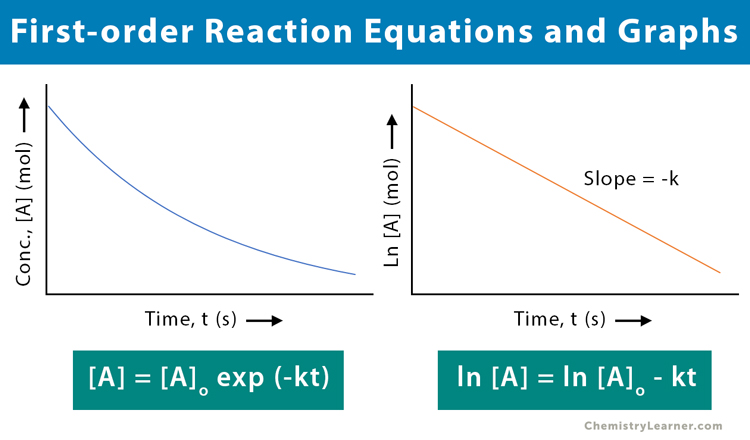
Half-Life of a First-Order Reaction Recall that for a first-order reaction the integrated rate law is given by. The half-life t_12 is a timescale on which the initial population is decreased by half of its original value represented by the following equation. The half-life of a first-order reaction does not depend upon the concentration of the reactant.
The half-life of a first-order reaction is 15 hours. However the half-life of a zero-order reaction increases as the initial concentration increases. A dfrac12 A_o After a period of one half-life t t_12 and we can write.
Since the reaction order is second the formula for t12. Where The half-life of a reaction is referred to as t 12 unit - seconds The initial reactant concentration is referred to as R 0 in molL. The half-life of a second-order reaction is given by the formula 1kR 0.
2λ 0693 λ. 453 t 1 2 0693 k. Ln 2 0693 kt12.
Thus for a first-order reaction each successive half-life is the same length of time as shown in. Graphical relations and half lives. T 12 0693 λ.
Substituting the values in the expression for the rate constant of half-life first-order reaction the. The half-life of a reaction is the time required for a reactant to reach one-half its initial concentration or pressure. As for all reaction orders the half-life for a zero-order reaction is inversely proportional to its rate constant.
However the half-life of a zero-order reaction increases as the initial concentration increases. λ 2 03465. Then half-life t 12 2λ.
What is the expression for Half-Life of a First Order ReactionHere I derive it from the integrated rate lawThe answer is t ln 2 kAsk me questions. For the first order reaction you can plug the definition of the half life into the concentration-time reaction to obtain a neat relationship. FraclnA_0A_tkt tfraclnA_0A_ttimesfrac1k.
T ½ 0693 k For a second order reaction 2A products or A B products when A B rate kA 2. T_12 frac1A_0k. Notice that the half life does not depend on the reactant concentration.
Half-lives of first order reactions. Find more Chemistry widgets in WolframAlpha. Plot the graph between Concentration Rate and Time for Zero Order Reactions.
Determining a half life. 2 0693 into the equation results in the expression for the half-life of a first-order reaction. For a zero order reaction A products rate k.
For first-order reaction we know that k 1t. Let the rate constant be λ. The first-order reaction half-life equation is given by k 2303 t l o g R 0 R From the definition of the half-life of a first-order reaction at t t12 and R R 02.

Zero Order Reactions Video Kinetics Khan Academy

Half Life Of A First Order Reaction Video Khan Academy

Determine The Half Life Of A First Order Reaction Youtube

Integrated Rate Laws Chemistry For Majors

Half Life Of A First Order Reaction Video Khan Academy

Half Life Of Zero Th 0th Order Reaction Derivation Youtube

Summary Of The Kinetics Of Zero Order First Order Ppt Download

Integrated Rate Laws Zero First Second Order Reactions Chemical Kinetics Youtube

Organic Chemistry Half Life And Shelf Life Of Second Order Reaction Chemistry Stack Exchange

How To Calculate Half Life Of A Second Order Reaction Chemistry Study Com
First Order Reaction Definition Examples And Equations

First Order Reaction Definition Example Half Life Period Chemist Notes

First Order Reaction Chemical Kinetics First Order Reaction Youtube

Calculate The Half Life Of A First Order Reaction From Their Rate Constants Given Below A 200 S 1 B 2 Min 1 C 4 Year 1

Chemical Kinetics Chapter 14 Chemistry The Central Science

First Order Reactions Chemistry Class 12 Iit Jee Main Advanced Neet Aipmt Askiitians Youtube